Will microbial manipulation sustain the ecological balance in shrimp (Penaeus monodon) hatcheries?
- Global styles
- MLA
- Vancouver
- Elsevier - Harvard
- APA
- Help

View/
Date
1998Page views
6,263ASFA keyword
AGROVOC keyword
Taxonomic term
Metadata
Show full item record
Share
Abstract
A shift in preferred methods employed to contain bacterial diseases in the hatchery phase of shrimp culture has resulted largely from the unsuccessful control by and deleterious effects of chemotherapy. Manipulation of hatchery microbial ecology has gained popularity, but for successful implementation, this niche-filling approach requires a thorough understanding of the epidemiology of bacterial diseases in the hatchery. This study examined the responses of Vibrio harveyi populations, (associated with luminescent vibriosis in shrimp larvae) to various physico-chemical factors and various hatchery components. Results showed that V. harveyi had a wider range of tolerance to environmental parameters than larvae of Penaeus monodon, such that control measures based on manipulation of these parameters might not be feasible. However, it was evident from the results that there were components in the shrimp hatchery environment that could be manipulated to control high populations of V. harveyi. The natural microflora of seawater, as well as the microbial flora associated with the diatoms Skeletonema costatum and Chaetoceros calcitrans negatively affected the survival of V. harveyi in experimental mixed cultures. The successful manipulation of such benign microbial components to compete with and exclude potential pathogens is necessary to sustain ecological balance in the shrimp hatchery environment.
Suggested Citation
Lavilla-Pitogo, C. R., Albright, L. J., & Paner, M. G. (1998). Will microbial manipulation sustain the ecological balance in shrimp (Penaeus monodon) hatcheries? In T. W. Flegel (Ed.), Advances in Shrimp Biotechnology : Proceedings to the special session on shrimp biotechnology, 5th Asian Fisheries Forum, 11-14 November 1998, Chiengmai, Thailand (pp. 185–192). Bangkok, Thailand: National Center for Genetic Engineering and Biotechnology.
Type
Conference paperISBN
9747578026Collections
- Conference Proceedings [300]
Related items
Showing items related by title, author, creator and subject.
-
Fish predation on mangrove-associated penaeids: The role of structures and substrate
Primavera, J. H. (Elsevier, 1997)The effect of habitat structure and substratum on predation of the greasyback shrimp Metapenaeus ensis (De Haan), white shrimp Penaeus merguiensis De Man and tiger shrimp Penaeus monodon Fabricius by sea bass Lates calcarifer Bloch and mangrove snapper Lutjanus argentimaculatus (Forsskal) was evaluated. The shrimp juveniles measured 6–15 mm in carapace length; fish measured 6.5–12.5 cm in standard length; structure types were pneumatophores of the mangrove Sonneratia griffithii Kurz and dried coconut leaf bracts; structure densities were 0, 32 and 98 pneumatophores per tank; and sediment particle sizes were pebbles, sand-granules and silt–sand. Predation on shrimp was significantly higher in controls or bare sand (48.7%) than among pneumatophores (29.9%), but not among leaf bracts (43.5%). Shrimp mortality was also significantly higher on bare sand (72.9%) compared to medium-density (54.2%), but not high-density (68.8%), pneumatophores. Fish predation on the burying shrimp M. ensis was affected by predator type but not by sediment size. The generally higher predation rates of snapper may be due to their habit of leaving unconsumed pieces of shrimp, whereas sea bass which devour whole prey require fewer shrimp to reach satiation. Moreover, the presence of structures did not affect sea bass behaviour of chasing prey among pneumatophores and under leaf bracts, but reduced predation by the relatively passive snapper. Predation rates among pneumatophores vs. control, and among medium-density pneumatophores vs. bare sand, were lower for P. monodon but not P. merguiensis. This may be related to the greater and more frequent use of (laboratory) shelters by juvenile tiger shrimp compared to white shrimp. The results demonstrate that the effective provision of shelter depends not only on structure type and density but on the behaviour of predator and prey as well. The use of mangrove structures (pneumatophores) by juvenile shrimp as refuge from predation is also documented for the first time. -
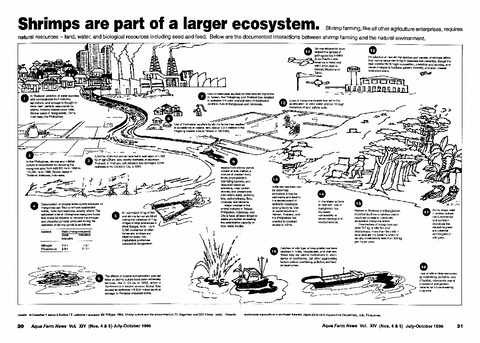
Shrimps are part of a larger ecosystem
Southeast Asian Fisheries Development Center, Aquaculture Department (Aquaculture Department, Southeast Asian Fisheries Development Center, 1996)The document illustrates the interactions between shrimp farming and the environment. -
Series: Aquaculture extension manual; No. 19
Prawn hatchery operations
Parado-Estepa, Fe D.; Quinitio, Emilia T. ; Borlongan, Emeterio L. (Aquaculture Department, Southeast Asian Fisheries Development Center, 1996-05)
The manual, an updated version of the 1984 SEAFDEC/AQD manual, presents the underlying principles and step-by-step instructions of prawn larval and post-larval rearing. The techniques described are not only applicable to Penaeus monodon, but may also be modified and applied to other penaeid and metapenaeid species. The following aspects are covered: Site selection; Life cycle; Hatchery design and planning; Hatchery operations; Diseases; and Economics.
; Borlongan, Emeterio L. (Aquaculture Department, Southeast Asian Fisheries Development Center, 1996-05)
The manual, an updated version of the 1984 SEAFDEC/AQD manual, presents the underlying principles and step-by-step instructions of prawn larval and post-larval rearing. The techniques described are not only applicable to Penaeus monodon, but may also be modified and applied to other penaeid and metapenaeid species. The following aspects are covered: Site selection; Life cycle; Hatchery design and planning; Hatchery operations; Diseases; and Economics.